( c) ATP synthesis rate in mock and altMiD51-expressing cells. Statistical significance was estimated using unpaired T-test. Results represent the mean value out of three independent experiments, with error bars corresponding to the standard error of the mean (s.e.m.). Cells were untreated, treated with a ROS inducer or a ROS inhibitor. ( b) ROS production in mock and altMiD51-expressing cells. Statistical significance was estimated using a two-way ANOVA with Tukey's post-hoc test (**p=0,004). Mean values of replicates are plotted with error bars corresponding to the 95% confidence intervals. The mitochondrial reserve capacity (maximal OCR- basal OCR) is an indicator of rapid adaptation to stress and metabolic changes. The balance of the basal OCR comprises oxygen consumption due to proton leak and nonmitochondrial sources. Mitochondrial function parameters were assessed in basal conditions (basal), in the presence of oligomycin to inhibit the ATP synthase (oxygen consumption that is ATP-linked), FCCP to uncouple the mitochondrial inner membrane and allow for maximum electron flux through the respiratory chain (maximal OCR), and antimycin A/rotenone to inhibit complex III (non-mitochondrial). ( a) Oxygen consumption rates (OCR) in HeLa cells transfected with empty vector (mock) or altMiD51 Flag. ***p<0.0005 (Fisher’s exact test) for the three morphologies between altMiD51(WT) and the other experimental conditions. Means of three independent experiments per condition are shown (100 cells for each independent experiment). ( d) Bar graphs show mitochondrial morphologies in HeLa cells. % of cells with the most frequent morphology is indicated: mock (tubular), altMiD51 WT (fragmented), altMiD51(LYR→AAA) (tubular), Drp1(K38A) (elongated).
In each image, boxed areas are shown at higher magnification in the bottom right corner. ( c) Confocal microscopy of mock-transfected cells, cells transfected with altMiD51 WT, altMiD51 LYR→AAA or Drp1 K38A immunostained with anti-TOM20 (red channel) and anti-Flag (green channel) monoclonal antibodies.

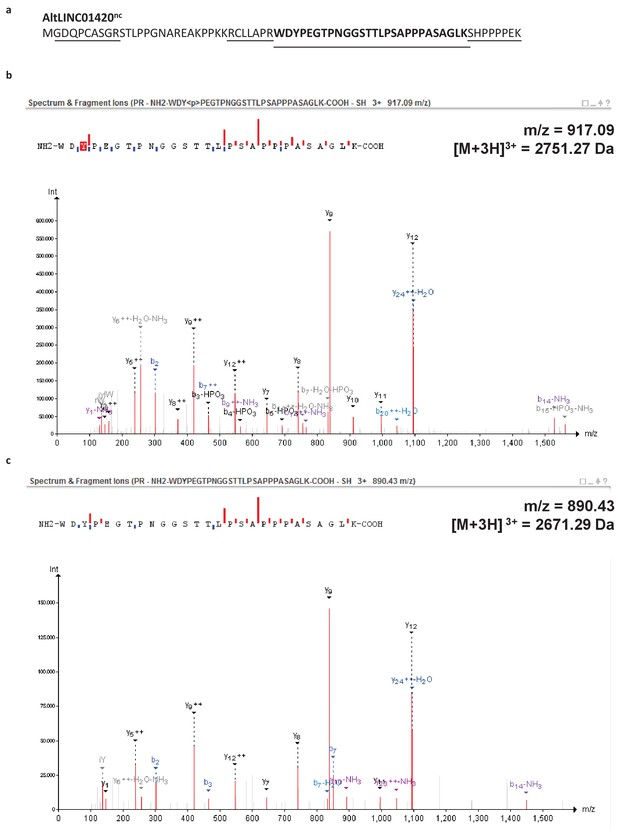
( b) Human HeLa cells transfected with empty vector (mock), a cDNA corresponding to the canonical MiD51 transcript with a Flag tag in frame with altMiD51 and an HA tag in frame with MiD51, altMiD51 Flag cDNA or MiD51 HA cDNA were lysed and analyzed by western blot with antibodies against Flag, HA or actin, as indicated. AltMiD51 amino acid sequence is shown with the LYR tripeptide shown in bold. ( a) AltMiD51 5’ coding sequence is located in exon two or the MiD51/MIEF1/SMCR7L gene and in the 5’UTR of the canonical mRNA (RefSeq NM_019008).+2 and+1 indicate reading frames. This suggests that 25.7% (35.7delete–10%) of these 889 altORFs undergo specific selection different from random regions in their CDSs with a similar length distribution. A random distribution would have implied a total of 10% (or 89) of altORFs in the extreme values. There are clearly substantial over-representations of extreme values (red signaling conservation DQ 0.95, and blue signaling accelerated evolution DQ 0.05) with 317 of 889 altORFs (35.7%). We show the absolute numbers (‘n’) and observed-to-expected ratios (‘O/E’) for each quantile. Expected quantiles of the differences (‘DQ’ columns) were identified and compared to the observed differences. We compared these differences to those generated based on five random regions in CDSs with a similar length as altORFs. PhyloPs for both altORFs and CDSs are based on third codons in the CDS reading frame, calculated across 100 vertebrate species. The plot contains 889 CDSs containing at least one fully nested altORF, paired with one of its altORFs selected at random (to avoid problems with statistical non-independence). We restricted the analysis to altORF-CDS pairs that were conserved from humans to zebrafish. Differences between altORF and CDS PhyloP scores (altORF PhyloP – CDS PhyloP, y-axis) are plotted against PhyloPs for their respective CDSs ( x-axis).
